
MEET NINA
Your Noninvasive
Neuromodulation Assistant

For patients who don’t want to take another pill and aren’t ready for surgery, NiNA* may be an ideal alternative to medication. NiNA uses proprietary technologies to optimally target and activate the saphenous while maintaining user comfort. This simple, discreet system for the treatment of OAB requires just 30 minutes of stimulation and is designed to deliver significant, durable symptom reduction all done in the convenience of your own home.
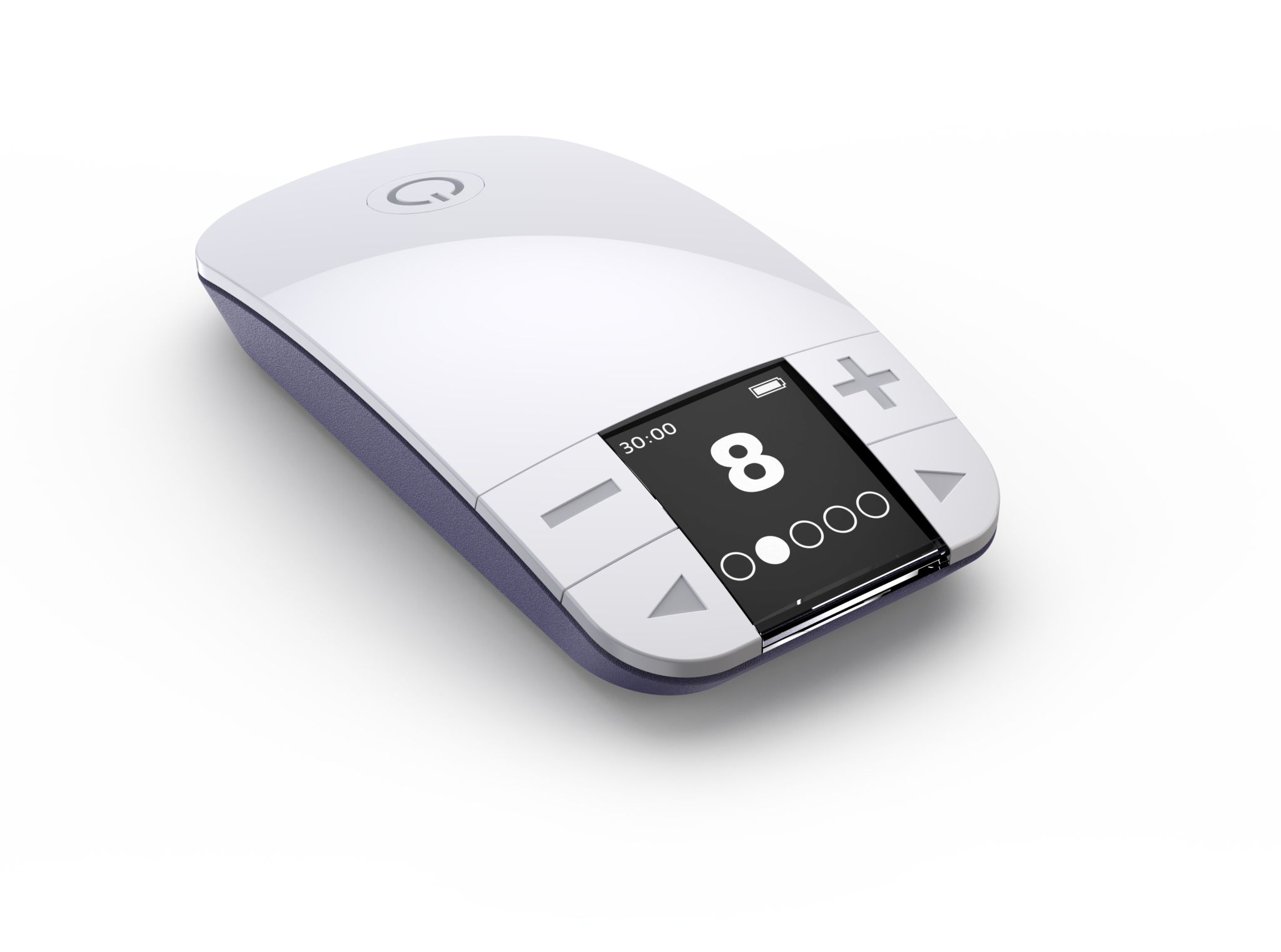
Drug-Free, Noninvasive, At-Home Therapy For OAB
85%
patients prefer NiNA to medications 3 to 1²
3 to 1
Physicians would prescribe NiNA to 85% of their OAB patients¹
Initial nSAFN clinical experience demonstrates efficacy on-par with published results for medications and other competing therapies.
Noninvasive Saphenous Neuromodulation (nSAFN) has been studied in 3 feasibility studies to date at 7 centers in the US and Canada.³
UUI reduction in UUI
responders³
78%
OAB responders³˒⁴
65%
85%
28%
UUI responders³
of patients were Dry³
Competitive Therapies
Medications
Percutaneous Tibial Neuromodulation (PTNS)
Sacral Neuromodulation (SNS)
Botulinum Toxin
Drawbacks
Significant side effects
Weekly office visits
Surgery required
Bladder wall injections
NiNA
NO serious side effects
Done at home
Completely noninvasive
Wearable device
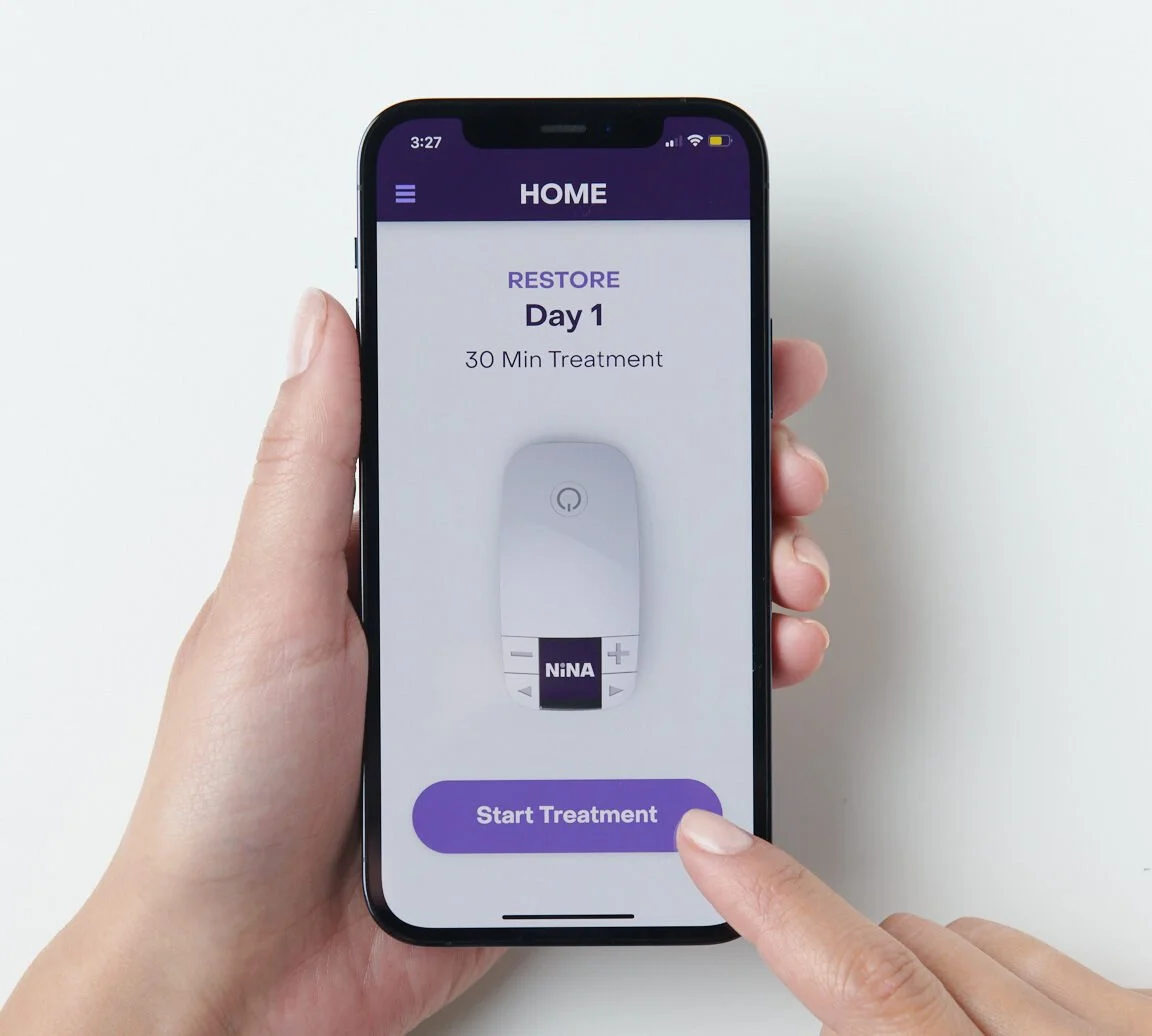
THE FUTURE OF NINA
Not only does NiNA offer revolutionary at-home neuromodulation therapy, but the introduction of the NiNA Care™ app⁶ will also add comprehensive educational content developed by world-renowned physicians– maximizing patient engagement and the potential for successful, durable outcomes.
An Intelligent Companion App and Ecosystem
¹Primary market research study, conducted by third party Q2-2021, data on file.
²Primary market research study, conducted by third party Q1-2021, data on file.
³Feasibility clinical outcomes, data on file.
⁴Defined as >=50% reduction or return to normal voiding. UUI = urinary urge incontinence.
⁵Brazzelli, M., et al. (2006). Efficacy and safety of sacral nerve stimulation for urinary urge incontinence: a systematic review. The Journal of Urology, 175(3 Pt 1), 835–841.
⁶NiNA Care is currently in development and has not received market authorization for sale in the US, Canada, or any other geography.
